Candace Owens: Coronavirus vaccination for children "a type of child abuse"
By nolanbarton // 2021-04-09
Tweet
Share
Copy
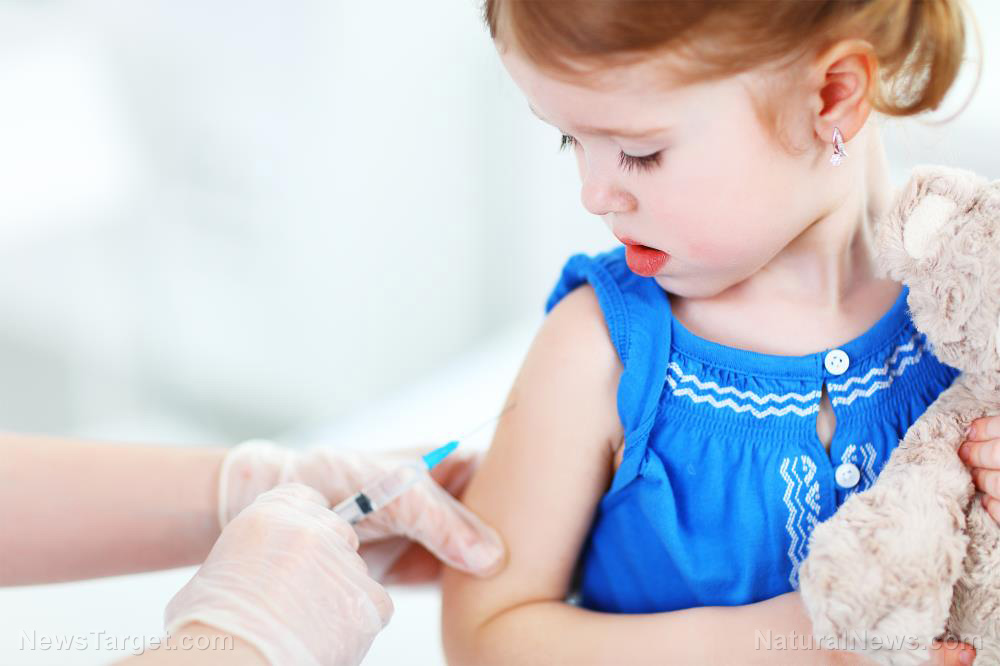
Conservative commentator and Blexit leader Candace Owens called the experimental coronavirus (COVID-19) vaccination for children "a type of child abuse."
On March 31, Owens tweeted: "Experimental vaccines on children who have an approximate 0% chance of dying from the virus for which they are being vaccinated against is a type of child abuse. Zero long term trials conducted. I just cannot understand parents that would allow their kids to be guinea pigs."
Big Tech doing the bidding of Big Pharma
Her tweet caught the attention of Twitter and promptly labeled it as "misleading." However, state-level data report about COVID-19 on children shows that the information in Owens's tweet is accurate. As of April 1, 0.00 to 0.003 percent of all COVID-19 cases involving children in the U.S. resulted in death, with 10 states reporting zero child deaths. According to the data published in the website of the American Academy of Pediatrics, there are 3,469,500 total child COVID-19 cases reported in the country. Of those cases, only 0.1 to 2.0 percent resulted in hospitalization. Owens posted two more tweets shortly after Twitter flagged her initial tweet. She reiterated her stance in the first tweet: "Twitter has unnecessarily slapped a label on my entirely truthful tweet. 1) There are no long term studies on the COVID-19 vaccines; 2) Children have an approximate 0% chance of dying from the virus; 3) I stand by my assessment that using children as lab rats is child abuse." In her next tweet, Owens said Twitter is doing the bidding of large pharmaceutical companies. She wrote: "Sticking bulls**t labels on tweets because Big Pharma tells you to is not going to make me get the vaccine. And it's sure as hell not going to transform my personal decision not to vaccinate my child. I'm not a sheep." (Related: If doctors offered truthful vaccine consent forms, no sane mom would get her child vaccinated.)COVID-19 vaccine for children is not a one-size-fits-all solution
The scientific community is facing a unique set of challenges in a bid to ensure that the COVID-19 vaccines are safe for children. Children are not involved in the original clinical trials because severe illness from COVID-19 is more common with adults, specifically older adults. Because a child's immune system is much different from an adult's, vaccines tend to require different dosage levels or formulations. Another thing to consider is that children's immune systems can vastly differ depending on how old they are. Thus, the COVID-19 vaccine for children couldn't be a one-size-fits-all solution. With more things to consider, medical trials involving children undergo stricter protocols than those involving adults.Pfizer-BioNTech announces successful vaccine trial for children
Pfizer-BioNTech recently announced that phase 3 of the trial for adolescents between the ages of 12 and 15 years old with or without prior evidence of SARS-CoV-2 infections demonstrated 100 percent efficacy and robust antibody responses. The trial included 2,260 children. "From the reports so far, it appears that not only does the vaccine elicit a good immune response, but it was very, very safe for the children and adolescents who received the vaccine," said Dr. Camille Sabella, a pediatric infectious disease specialist who was not involved in the trial. In the trial, 18 cases of COVID-19 were observed in the placebo group (1,129 children) versus none in the vaccinated group (1,131 children). The results showed that the vaccine was equally effective at producing an antibody response in younger children as compared to the teens and young adults between the ages 16 and 25 years old from an earlier trial. Dr. Sabella said that the results from the phase 3 trial will need to undergo peer review and be presented to the Food and Drug Administration (FDA) to get an authorization for emergency use. Pfizer-BioNTech reported that the side effects were consistent with those reported during their earlier trial with 16 to 25 year old participants. According to the company, all participants in the trial will be monitored for long-term protection and safety for an additional two years after their second dose. The company also announced that it has started a global study to evaluate the safety, tolerability and effectiveness of their COVID-19 vaccine in children aged 6 months to 11 years old.Other pharmaceutical companies conducting their own studies
Moderna has also started its study with children under 12 a few weeks ago. The company is expecting around 6,750 healthy children in the U.S. and Canada to participate in the trial. Earlier in March, Johnson & Johnson said it is likely to have a COVID-19 vaccine for children under 18 by September. The company is also planning to start testing adolescents between the ages of 12 and 17 and then down to infants. Meanwhile, a trial of the Oxford-AstraZeneca COVID-19 vaccine on children has temporarily stopped while the United Kingdom's drug regulator – Medicines and Healthcare products Regulatory Agency (MHRA) – investigates the vaccine's possible link with rare blood clots in adults. Oxford University said in a statement that there were "no safety concerns" in the trial, but acknowledged fears over a potential link to clots by saying that it was awaiting additional data from Britain’s Medicines and Healthcare products Regulatory Agency (MHRA) before restarting the study. (Related: SACRIFICE THE CHILDREN: Oxford Vaccine Group recruits children for coronavirus vaccine trials.) "Parents and children should continue to attend all scheduled visits and can contact the trial sites if they have any questions," Oxford stated.Vaccination should not be a prerequisite to have children back in school
Children have generally experienced milder problems from COVID-19 and most of them don't have significant symptoms. However, some children have become severely ill from acute COVID-19 or from post-COVID-19 Multisystem Inflammatory Syndrome in Children (MIS-C). Health experts also say children can still transmit the virus. That's why they deem it important to protect the children from the disease. But Dr. Sabella does not believe that vaccinating children should be a prerequisite to have them back in school. "We've learned over the last few months that children can be in school safely as long as all the precautions are taken to keep them safe with social distancing, masking and proper hand hygiene. Many school districts have done a wonderful job with this. Vaccination will just be one more step in helping people, especially parents, feel more comfortable with sending kids back to school," Dr. Sabella said. "It's important for kids to do the things that they like doing. It's good for their physical and mental health. However, we still need to take all of the precautions that are in place." Follow Immunization.news for more news and information related to coronavirus vaccines. Sources include: LifeSiteNews.com Health.ClevelandClinic.org Aljazeera.comTweet
Share
Copy
Tagged Under:
mental health bad medicine twisted Big Pharma Twitter Child abuse infertility depopulation pandemic outbreak Blood clots lab rats Big Tech guinea pigs badscience badhealth badmedicine big tech censorship coronavirus COVID-19 vaccine vaccine trial
You Might Also Like
By News Editors // Share
Whole Foods ushering in Mark of the Beast with new palm-scanning payment system
By Ethan Huff // Share
Why are certain 5G cell tower components labeled “COV-19?”
By Ethan Huff // Share
Recent News
The Home Front: When the calorie becomes the last honest currency
By bellecarter // Share
Iran's Phoenix: The Military Industrial Resurrection That Washington Cannot Stop
By garrisonvance // Share
EU and U.S. launch strategic stockpile to break China's rare earth stranglehold
By jacobthomas // Share