The alloys used in the process could also be recycled
The MIT researchers found that the gallium-indium mixture penetrates and reaches the interior grains of the material samples. They noted, however, that the rate of penetration varies depending on the other elements present in the sample. Pure aluminum and those with silicon had the same permeation rate, but the eutectic mixture penetrated samples with magnesium at a slower rate. Study co-author Laureen Meroueh mentioned the different benefits of using the aluminum-water process to produce hydrogen. She said: "You don't have to apply any energy for the gallium-indium eutectic [mixture] to work its magic on aluminum and get rid of that oxide layer. Once you've activated your aluminum, you can drop it in water and it'll generate hydrogen." (Related: Can we find a cleaner way to extract hydrogen from water?) She also noted that the eutectic mixture does not start a chemical reaction with the aluminum; it instead "physically moves around" in between the aluminum grains. Meroueh also pointed out that the gallium and indium eutectic mixture can be recovered, given that both are expensive and in short supply. "At the end of the process, I could recover all of the gallium and indium I put in and use [them] again," she said. (Related: Bacteria can recover precious metals from old batteries for use in new ones.) Hart, Meroueh and co-author Thomas Eagar observed that aluminum scraps that contained silicon and magnesium exhibited a different property than the rest of the samples. Often found in recycling streams, aluminum mixed with the two elements yielded a moderately strong yet corrosion-resistant material. Thus, they hypothesized that the presence of magnesium silicide – the substance formed when the two alloys combine – blocked the eutectic mixture from penetrating. The hypothesis was proven true after coated aluminum scraps with silicon and magnesium were observed under a scanning electron microscope. The magnesium silicide deposits acted as barriers that prevented the gallium-indium mixture from permeating, even though the samples were already treated for 48 hours. Thus, the MIT researchers indicated that with the incomplete process, a lengthy pre-treatment period for aluminum mixed with silicon and magnesium would be critical for maximizing its hydrogen yield. Less than a year earlier, Belgian scientists managed to develop a solar panel that produced hydrogen aside from energy. The researchers at KU Leuven led by professor Johan Martens said the panel also made use of water – in the form of moisture in the air – to generate hydrogen. They added that the resulting hydrogen can then be used for home heating systems. A demonstration of the panel showed that it can produce substantial amounts of hydrogen. Large quantities of hydrogen bubbles appeared as soon as the prototype was exposed to the sun. Researcher Jan Ronge, who was a member of Martens's team, said of the solar panel: "Twenty of these … produce enough heat and electricity to get through the winter in a very well-insulated house, and still have electricity left over." NewEnergyReport.com has more articles about hydrogen production with only the help of water. Sources include: Phys.org Pubs.ACS.org CleanTechnica.comIsraeli study shows the human immune system is stronger than vaccines
By Mary Villareal // Share
Pomegranate peel has protective effects against enteropathogenic bacteria
By Ralph Flores // Share
Lantana grisebachii found to prevent bioaccumulation of arsenic in rat splenocytes
By Ralph Flores // Share
Mysterious jellyfish-like object filmed entering Earth’s atmosphere
By Mary Villareal // Share
House Advances $8 Billion Ukraine Aid Package Amid Congressional Pushback on Foreign Policy
By garrisonvance // Share
Trump Signs Order for Government Reviews of Advanced AI Models Before Release
By chasecodewell // Share
Report: Improper Obamacare Payments Could Reach $25B in 2026
By morganverity // Share
Meta Accused of Fostering Ethnic Enclaves at Headquarters
By edisonreed // Share
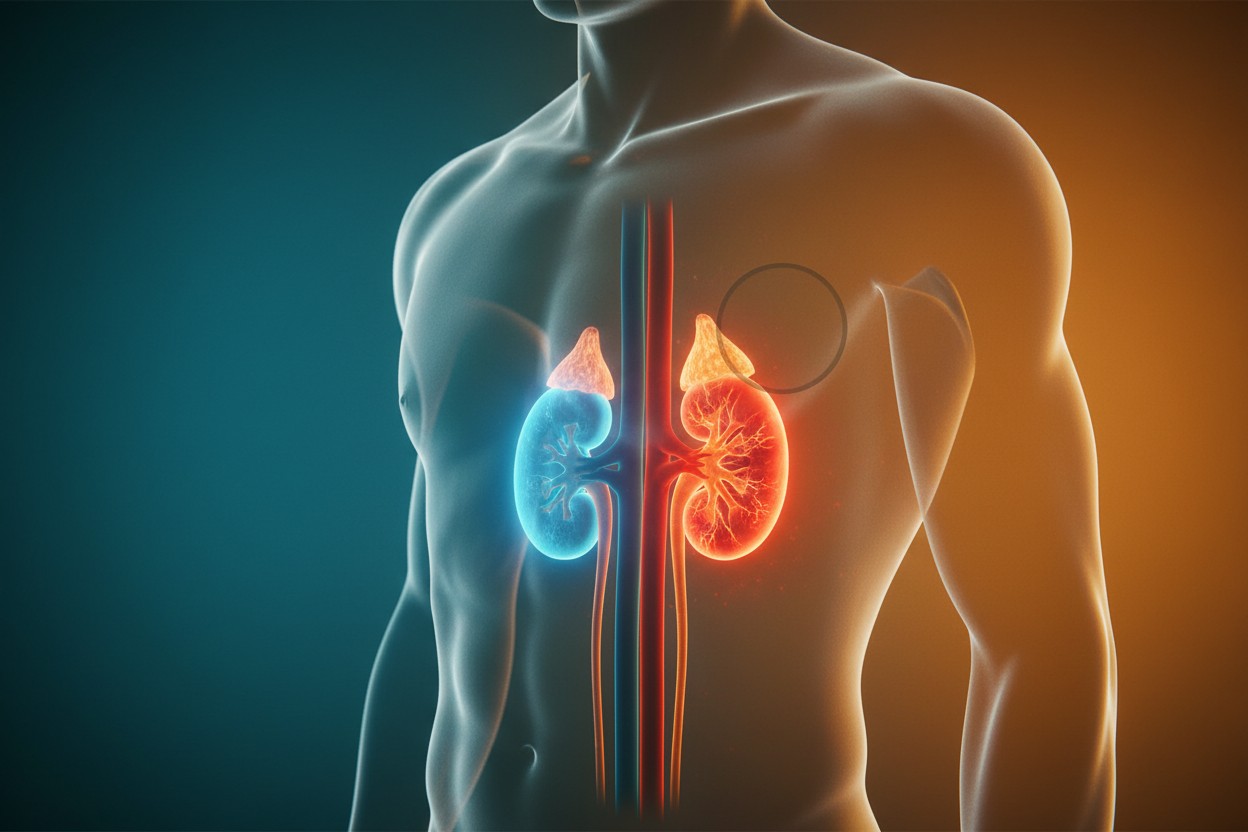
Nearly 800 Million Adults Have Chronic Kidney Disease, With Most Unaware
By morganverity // Share